RNA-binding proteins in brain disease
RNA-binding proteins play central roles in how cells manage RNA, including splicing,
localisation, stability and translation. In the nervous system, these processes are
especially important because neurons are highly specialised, long-lived cells with
complex RNA demands.
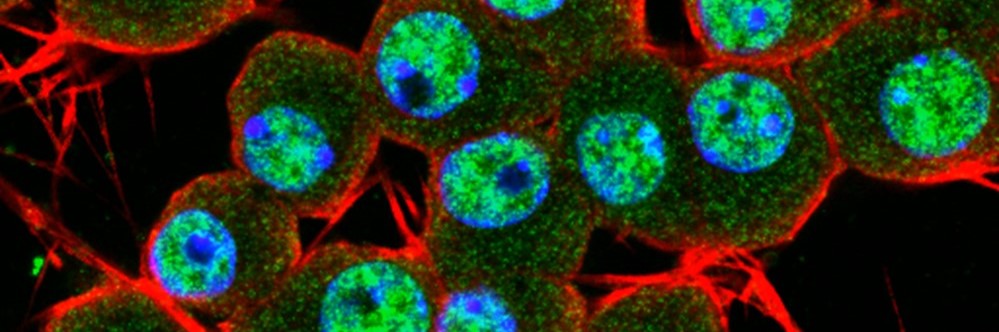
Our work focuses on understanding how disruption of proteins such as TDP-43 and related
RNA-binding proteins affects neuronal systems in conditions including motor neurone
disease, frontotemporal dementia and related disorders.
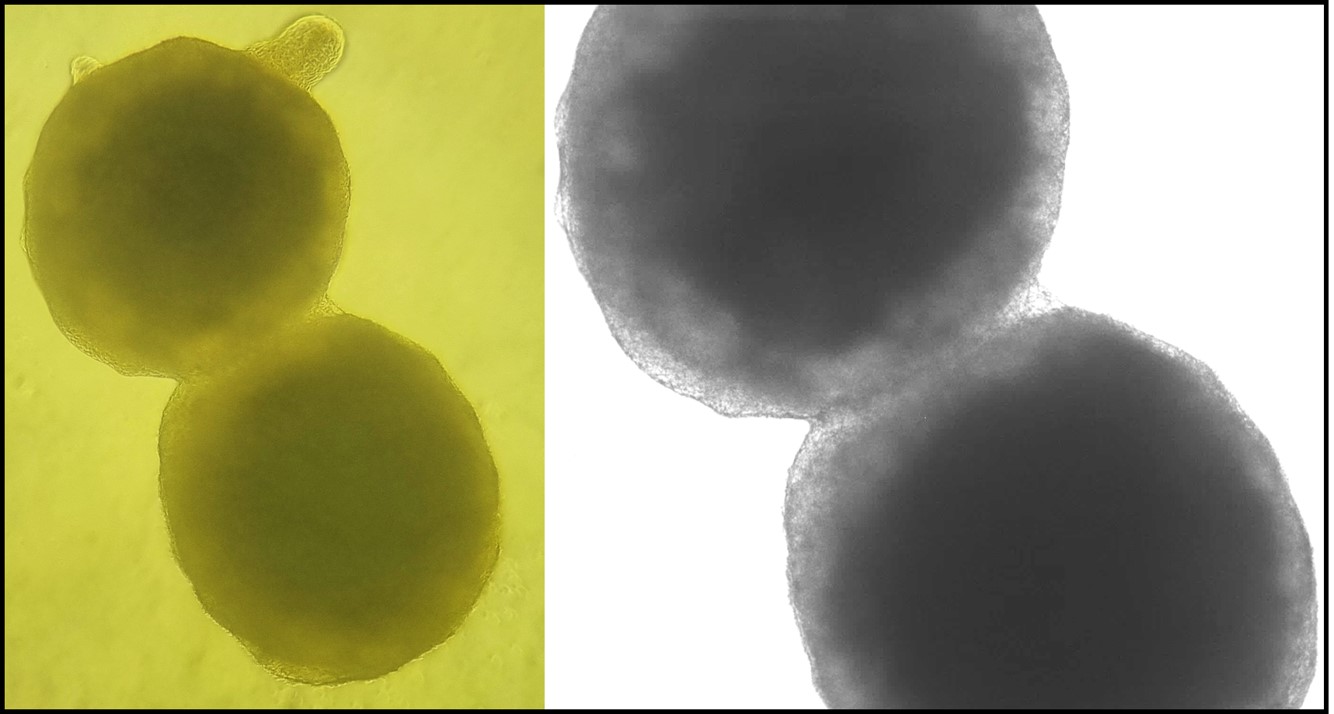
Human stem-cell models of the nervous system
A central part of the lab’s approach is the use of human stem-cell-derived models.
These include 2D neuronal cultures as well as more complex 3D systems such as forebrain
organoids and assembloids.
Using a combination of systems helps us study disease mechanisms at different levels of
complexity, from controlled discovery work to more tissue-like biological contexts.
Transcriptomics, splicing and isoform biology
One of the main ways we study disease is by looking closely at RNA itself. We use
transcriptomic approaches to understand how gene expression changes when RNA-binding
proteins are disrupted.
This includes studying changes in overall gene activity, RNA splicing, cryptic exon
inclusion and isoform usage using bulk, single-cell and long-read sequencing strategies.
Early mechanisms and therapeutic discovery
We are particularly interested in the earliest disease-relevant changes that occur after
RNA regulation is disturbed. Understanding these early events can help us prioritise
which pathways matter most and which cell states appear most vulnerable.
Over time, this helps create a framework for biologically informed therapeutic discovery.